Article
Incorporating
Clinical Perspectives
in modeling via Delphi
Incorporating clinical perspectives to inform cost-effectiveness analysis (CEA) has become of increasing importance with trends in biopharmaceutical development. Between 1990 and 2010, development of therapies for rare diseases grew by 4X based on US Food and Drug Administration (FDA) orphan-drug designations.1 As clinical evidence is often limited for rare diseases, perspectives of rare disease clinical experts are critical for development of appropriate disease models.
Additionally, CEA requires modeling long-term outcomes associated with quality of life and costs, while many novel biopharmaceutical interventions are granted regulatory approval based on shorter-term “surrogate” endpoints (biomarkers or other intermediate outcomes, predictive of longer-term outcomes). Over the last 30 years, >50% of new drug and biological treatment approvals by the US FDA, European Medicines Agency (EMA), and Pharmaceuticals and Medical Devices Agency (PMDA) in Japan have been reported to be based on surrogate-endpoint data from clinical studies.2 Where evidence is lacking to characterize the relationship between a surrogate endpoint and long-term outcomes, clinical perspective may help characterize this association.3
The Delphi method provides a systematic approach for evaluating perspectives of multiple stakeholders (e.g., clinical experts), and thus can facilitate development of a clinically accurate disease model for HTA submissions. It is an iterative, structured, group-based method for expert elicitation and stakeholder engagement. Expert participants provide their opinions in response to sequential “rounds” of questions. Each round is informed by the group’s responses from the previous round, encouraging participants to reassess, alter, and/or further develop their opinions.4Responses are anonymous to ensure that no individual dominates the process.5,6 In healthcare-oriented research, “modified Delphi” panels are common, involving two rounds with an intermediate review and discussion of anonymized responses.5A modified Delphi panel typically follows this structure, although the term has not been officially defined;5 it is generally understood to refer to a process following the RAND/UCLA Appropriateness Method (RAM).6,7 In recent years, “e-Delphi panels” have been used increasingly,8,9 which generally take the form of a modified Delphi panel, but the discussion is held through an online forum.10
To inform development of a CEA for a novel therapy for treatment of a rare disease called phenylketonuria (PKU), Medicus recently conducted an eDelphi study.11 The study aimed to identify areas of consensus (or lack thereof) for disease modeling. An international sample of experts with experience managing patients with PKU was obtained to understand potential for heterogeneity in clinical perspectives across countries. Figure 1 summarizes the steps taken to elicit perspectives from clinical experts to inform a CEA disease model.
Figure 1: Modified Delphi Process
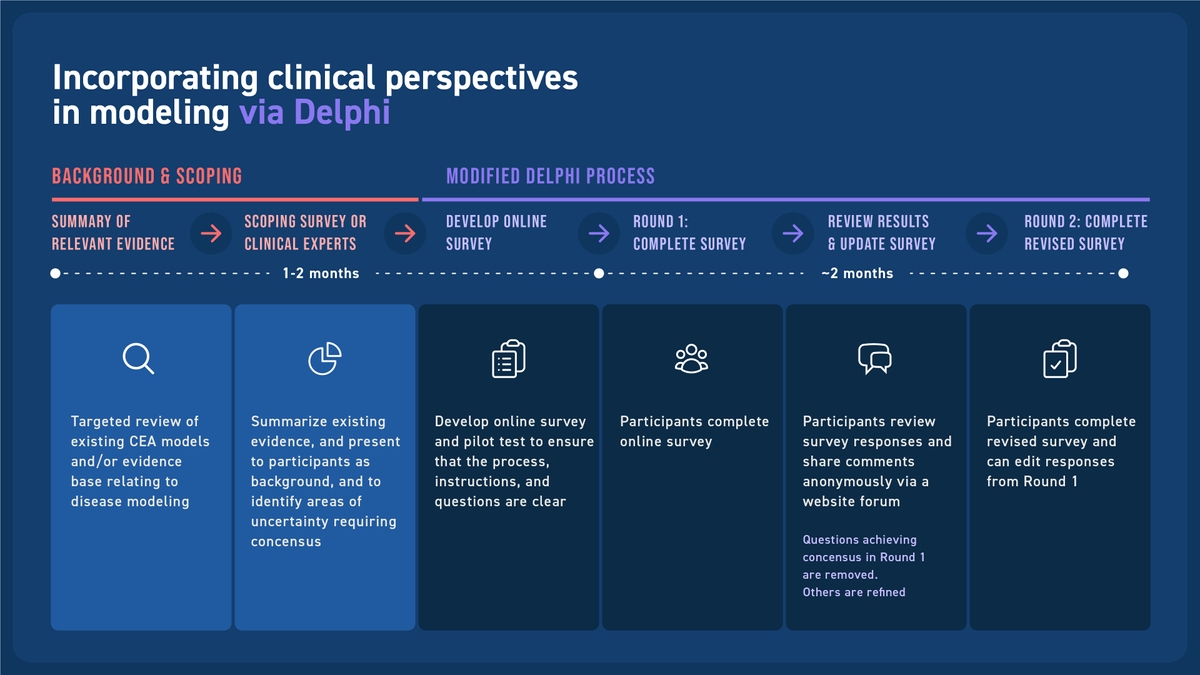
As was the case with this example, an e-Delphi study can effectively facilitate incorporation of current clinical thinking in disease modeling for HTA. It can provide a fast (3-4 months) approach for developing a defensible, documented clinical basis for disease modeling in CEA. This may be particularly important for capturing the value for cutting edge therapies with surrogate endpoints or therapies for rare diseases, where existing disease models are less established.
1 Miller KL, Fermaglich LJ, Maynard J. Using four decades of FDA orphan drug designations to describe trends in rare disease drug development: substantial growth seen in development of drugs for rare oncologic, neurologic, and pediatric-onset diseases. Orphanet J Rare Dis. 2021;16(1):265.
2 Ciani O, Manyara AM, Davies P, Stewart D, Weir CJ, Young AE, et al. A framework for the definition and interpretation of the use of surrogate endpoints in interventional trials. EClinicalMedicine. 2023;65:102283.
3 CDA-AMC. White Paper - Surrogate endpoints in cost-effectiveness analysis for use in health technology assessment. January 2025. Available at: https://www.cda-amc.ca/sites/default/files/MG%20Methods/surrogate-endpoints-report.pdf.
4 Keeney E, Thom H, Turner E, Martin RM, Sanghera S. Using a Modified Delphi Approach to Gain Consensus on Relevant Comparators in a Cost-Effectiveness Model: Application to Prostate Cancer Screening. Pharmacoeconomics. 2021;39(5):589-600.
5 Khodyakov D. What is Delphi? March 25, 2024. URL: https://www.linkedin.com/pulse/what-delphi-dmitry-khodyakov-hmhwc/.
6 Khodyakov D, Grant S, Kroger J, bauman m. RAND Methodological Guidance for Conducting and Critically Appraising Delphi Panels2023.
7 Kathryn F. The Rand/UCLA appropriateness method user's manual: Santa Monica : Rand, 2001.; 2001.
8 Délétroz C, Del Grande C, Amil S, Bodenmann P, Gagnon MP, Sasseville M. Development of a patient-reported outcome measure of digital health literacy for chronic patients: results of a French international online Delphi study. BMC Nurs. 2023;22(1):476.
9 Graf M, Kim E, Brewer I, Hernandez J, Chou JW, Cirillo J, et al. Expert consensus established around flexible, individualized migraine treatment utilizing a modified Delphi panel. Headache. 2023;63(4):506-16.
10 Khodyakov D, Grant S, Denger B, Kinnett K, Martin A, Peay H, Coulter I. Practical Considerations in Using Online Modified-Delphi Approaches to Engage Patients and Other Stakeholders in Clinical Practice Guideline Development. The Patient - Patient-Centered Outcomes Research. 2020;13(1):11-21.
11 Zhang R, Chakrapani A, Hamazaki T, Lah M, Muntau AC, Ruebel DJ, Vijay S, Zori RT, Feillet F, O'Connell T, Woolley JJ, Crowell M, Teng Y, Tomazos I. An iterative survey of phenylketonuria (PKU) medical experts to inform health economic modeling methods. ISPOR Europe 2024, Barcelona, Spain. Value in Health, Volume 27, Issue 12, S2.